Scientific question
Our model of choice is the visual system of Drosophila melanogaster, because many cell types have been mapped and are accessible for genetic manipulations at the single cell level. Flies have almost crystalline compound eyes, that are composed of around 750 units, the ommatidia. Each facet holds eight types of light-sensitive photoreceptors. Their axons project into the underlying optic lobe, consisting of four areas, the lamina, medulla, lobula plate and lobula. The visual system contains a multitude of neuron subtypes whose neurites are arranged in highly regular, intricate patterns of columns and layers. Neurons are closely associated with another diverse cell population, the glia.
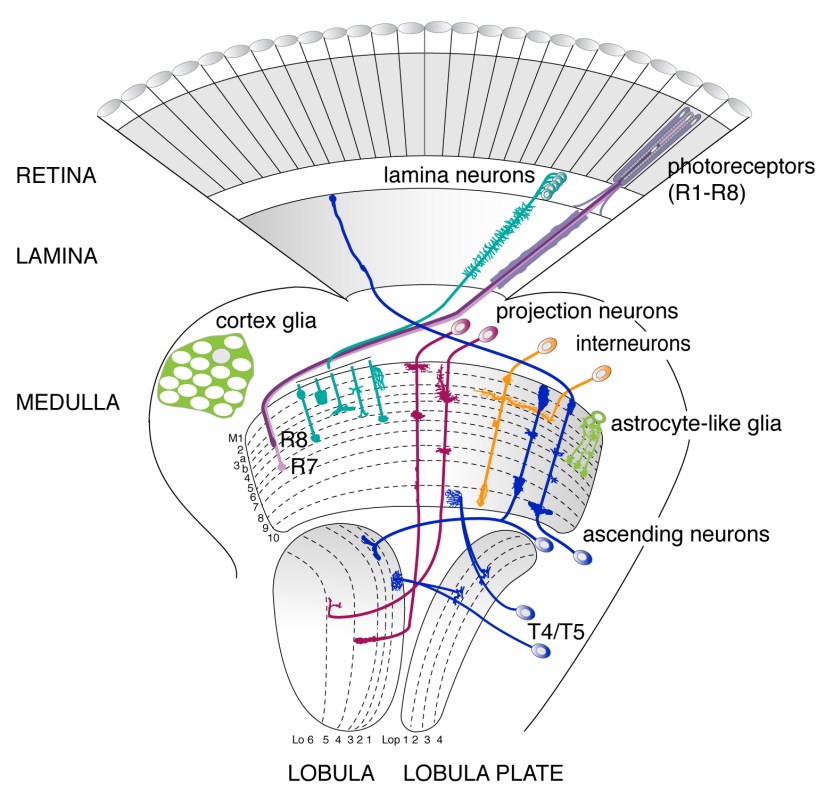
Adult visual system Drosophila.
Drawing: I. Salecker
During development, every neuron and glial cell needs to be born, adopt a specific identity, find its correct location and extend branches with specific shapes. Neurons not only have to connect with their correct synaptic partners but also interact with glia to form functional circuits. Projects in our lab seek to understand how specific developmental steps during neural network formation are orchestrated on a cellular and molecular level. Each finding is part of a fascinating jigsaw puzzle. A better understanding of these mechanisms in the growing brain will help to provide insights relevant for determining the causes of neurodevelopmental and neurodegenerative disorders at long term.
Research aims
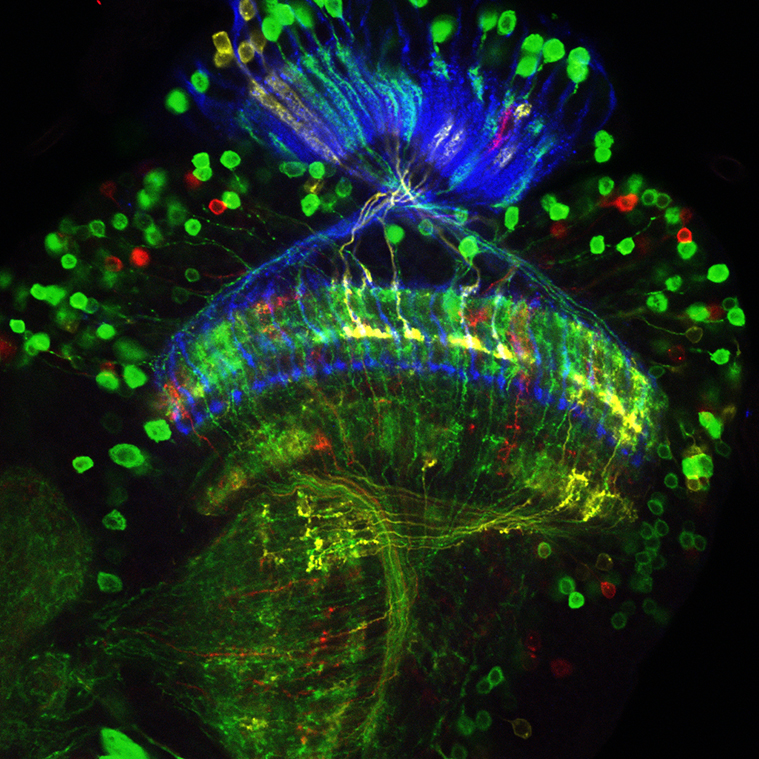
Netrin-Gal4 labeled neurons.
Image: D. Hadjieconomou
SEQUENTIAL GANGLION AND LAYER SELECTION
How higher-order neurons in the visual system make binary decisions to first choose the correct area over another and then a specific synaptic layer within this region, remains poorly understood. Using our earlier studies of photoreceptor axon targeting as an entry point (Timofeev, Joly et al., 2012), this project seeks to unravel the function of candidate molecules in mediating sequential decisions within the same neuron subtype.
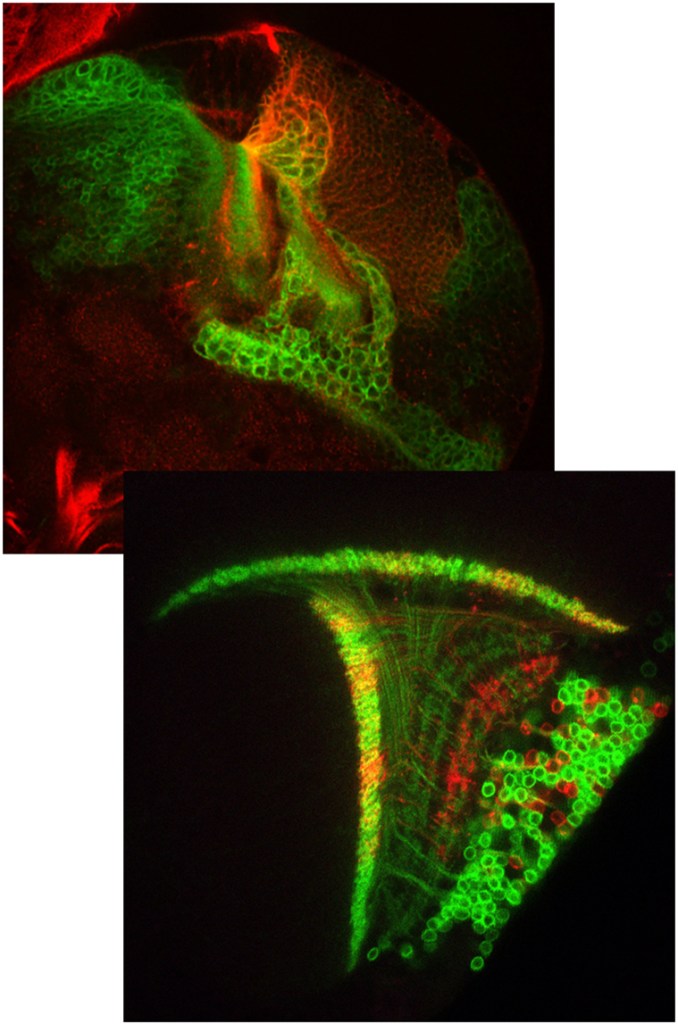
T4/T5 neuron development.
Images: H. Apitz
LAYER FORMATION OF MOTION DIRECTION-SELECTIVE NEURONS
Our previous studies of motion direction-selective T4/T5 neurons uncovered the mechanisms controlling their early development. This includes a unique mode of neurogenesis with migratory progenitors and a two-step specification process of neural stem cells (Apitz and Salecker, 2015, 2016, 2018). Our aim is to further understand how these neurons acquire their subtype identities and how they organize their neurites into four distinct synaptic layers in interaction with partner neurons.

Astrocyte in the adult medulla.
Image: B. Richier
ASTROCYTE MORPHOGENESIS AND ROLE IN CIRCUIT FORMATION AND FUNCTION
We previously found that the transmembrane protein Lapsyn controls branch morphogenesis of astrocyte-like glia in the medulla (Richier, de Miguel Vijandi et al., 2017). Our current studies ask how astrocytes acquire their distinct branching patterns in interactions with neurons and how they influence circuit function. Our entry point is the identification of binding partners of Lapsyn on the neuronal and glial side using mass spectrometry.
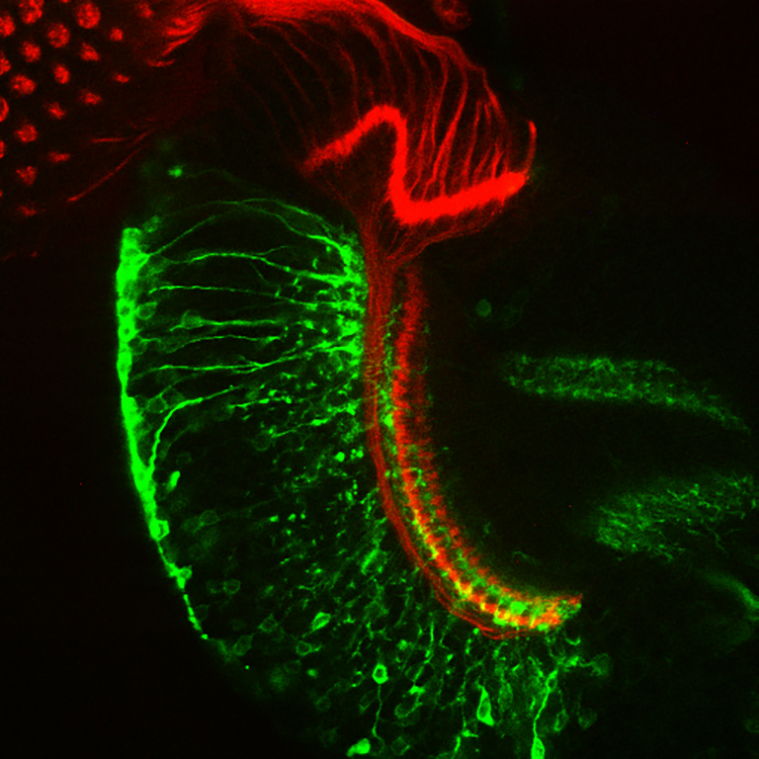
Remodeling medulla neurons.
Image: I. Salecker
DEVELOPMENTAL NEURITE REMODELING DURING LAYER FORMATION
Neurite remodeling offers plasticity in refining basic wiring patterns. Our ongoing studies revealed that some neuron subtypes in the medulla undergo transitions reminiscent of neurite remodeling. Our goal is to pinpoint the role of glia in mediating this so far unexplored developmental step during layer formation in the visual system and to identify the key molecular determinants using transcriptomics approaches. We hypothesize that this step is crucial in the context of topographic mapping.
